Data relations are at the core of Life Science discovery. Powered by a relational data model Siren turns traditional “dashboard” analytics into something completely new combining instruments that were previously totally disconnected or simply unavailable. Watch it in action now.
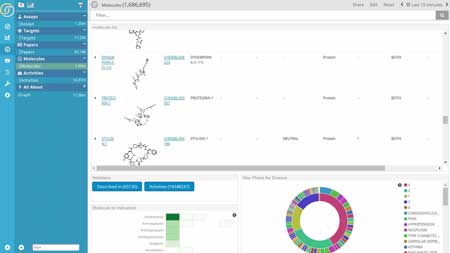
Whether examining the inter-dependency of data to avoid skewing analysis of laboratory results or searching for all data associated with a given molecule across in-house and external sources, researchers want to gain more insight into their data to help make better decisions. They are looking for ways to quickly get answers to their questions without relying on it.
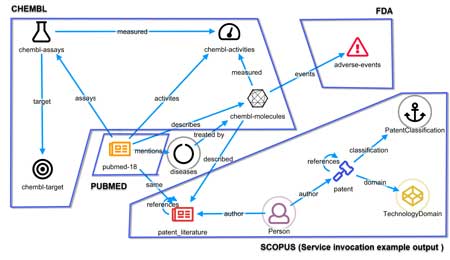
Relations among data sets are at the core of discovery in Life Sciences. Marrying real-time data with historical data, both structured and unstructured, across different sources is critical to gaining a competitive advantage. The Siren Platform™ provides relational data model driven drill downs, empowering domain experts with the ability to seamlessly navigate across related records through the combination of relational dashboards and state-of-the-art link analysis.

Knowing what problems occurred with clinical trials in the past helps pharmaceutical companies to better plan them in the future to accelerate the drug development process. Quality and Audit leaders want to rapidly see the relationship between their providers, audits, investigations, findings, and CAPAs to drill down to root causes that can be addressed. Being able to get this level of visibility across multiple trials gives them the insight to drive efficiency in planning future trials.
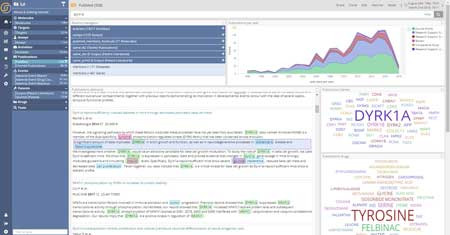
Every big data vendor in Life Science typically showcases screenshots that might resemble the one we show here – i.e. with focused entity extractions and analytics. But in Siren this is just the first step: start from Textual data search and drilldown and use as a way to navigate and drilldown on other, non textual, datasets you might have. Or vice versa!
Have great NLP in house? Siren can be configured to work with that, leveraging and augmenting the investments you have already made.

The use of medical devices and wearable technology is exploding in life sciences. Patient adoption is growing due to the improved quality of life opportunities that these technologies offer. These data flows are challenging the traditional infrastructure of pharmaceutical companies that were not designed for streaming data in large volumes. A platform that can handle structured and unstructured data that can be either static or real-time allows the data from these sources to be combined with the rich treasure trove of data that already exists to develop new insights.
Our experts can show you exactly how to leverage your data to uncover powerful insights!